The mass of an electron is 9.1 X 10^-31 kg. If its K.E is 3.0 X 10^ -25 J. calculate its wavelength. - YouTube
Given: The mass of electron is 9.11 x 10^-31 kg, Planck constant is 6.626 x 10^-34 J s, - Sarthaks eConnect | Largest Online Education Community

If `m,e,epsilon_(0) h` and `c` denote mass ,electron , change of electron, plank \'s constant and - YouTube

SOLVED: Planck's constant is h = 6.626X 10-34J . vacuum is c =3 X 108@ 4.136 x 10-I4eV . S; Speed of light in Elementary charge: € = 1.60 X1O-19 C; mass

Intramolecular Electron-Transfer Rates in Mixed-Valence Triarylamines: Measurement by Variable-Temperature ESR Spectroscopy and Comparison with Optical Data | Journal of the American Chemical Society

Uncertainty in the position of an electron (mass = 9.1 × 10^-31kg) moving with a velocity 300ms^-1 accurate upon 0.001

Synthesis, Electronic Structure, and Electron Transfer Dynamics of (Aryl)ethynyl-Bridged Donor−Acceptor Systems | Journal of the American Chemical Society
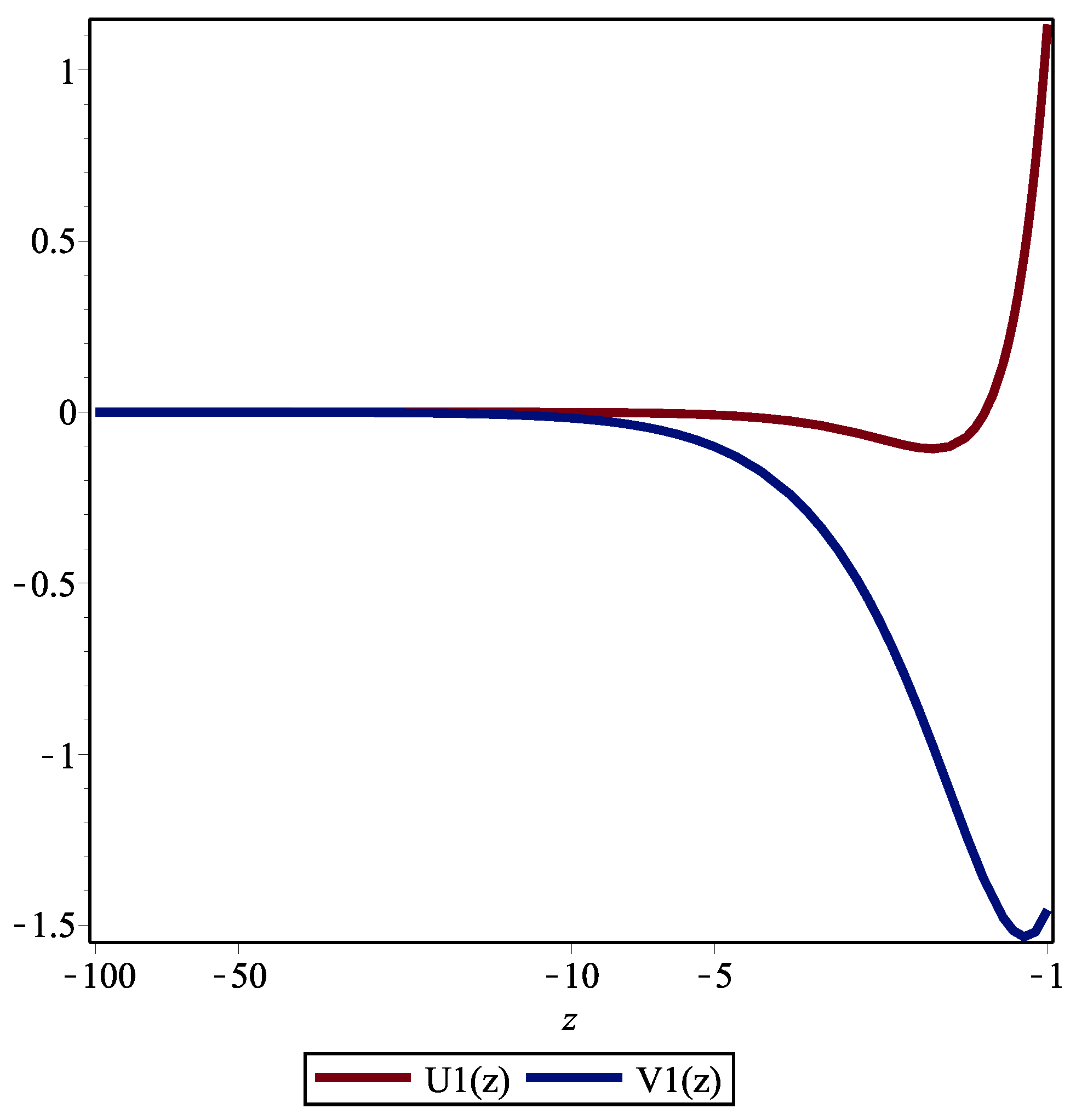
AppliedMath | Free Full-Text | Approximate Nonlocal Symmetries for a Perturbed Schrödinger Equation with a Weak Infinite Power-Law Memory

Given: The mass of electron is `9.11 � 10^(�31)`Kg Planck constant is `6.626 �10^(�34)` is:- - YouTube
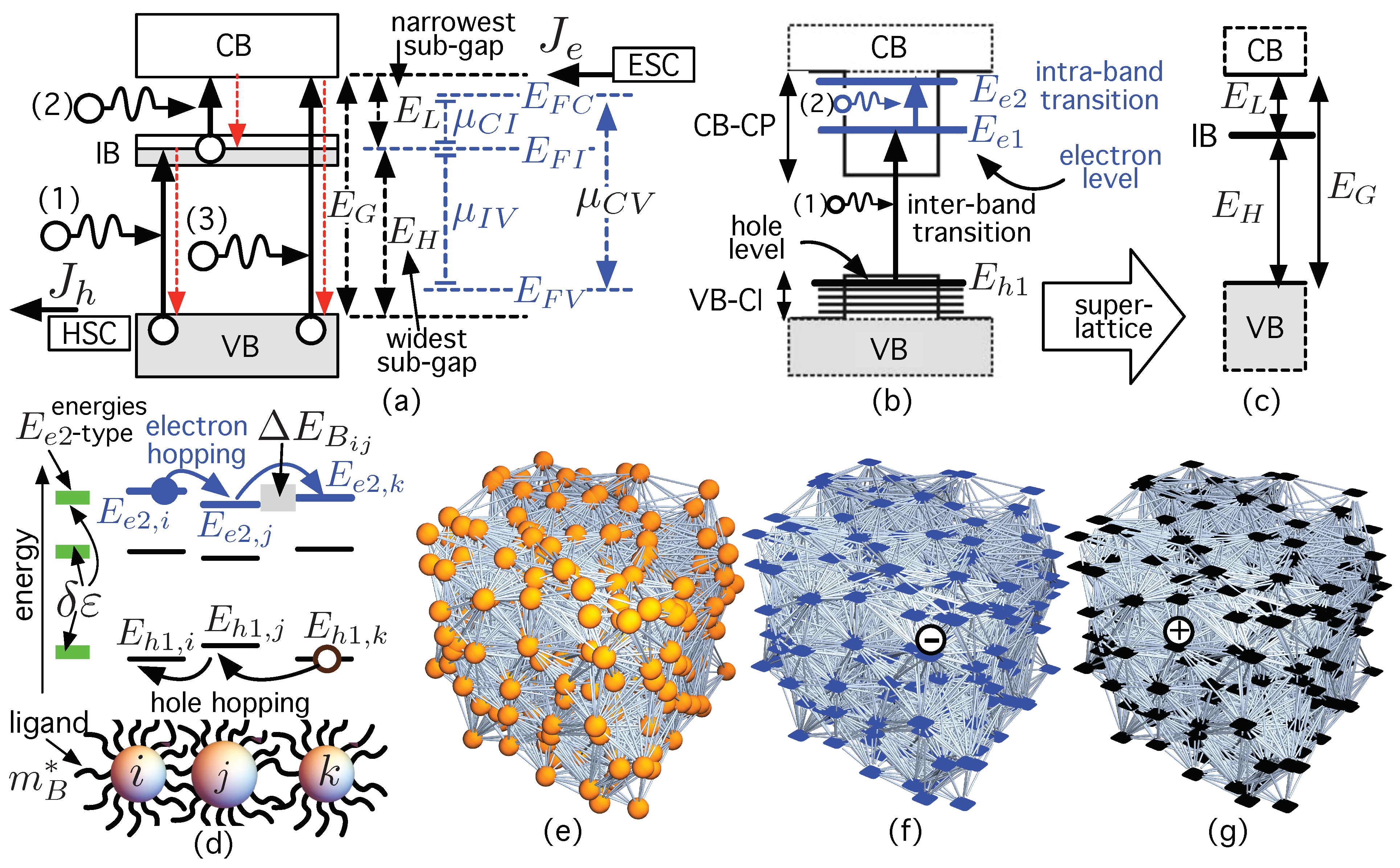
IJMS | Free Full-Text | Carrier Transport in Colloidal Quantum Dot Intermediate Band Solar Cell Materials Using Network Science